Beta-lactam Antibiotics
Beta-lactam Antibiotics
Bacterial cell walls contain peptidoglycan, a polymer consisting of sugar chains cross-linked by short peptide chains. This structure provides mechanical strength and protection against osmotic lysis.
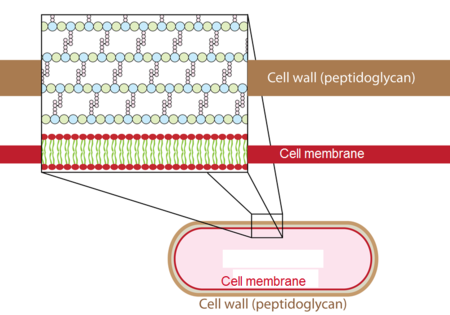
Beta-lactam antibiotics target penicillin-binding proteins (PBPs), which are enzymes responsible for the final steps of peptidoglycan synthesis. These PBPs include:
- Transpeptidases – responsible for cross-linking peptidoglycan strands.
- Carboxypeptidases & Endopeptidases – involved in remodeling and maintaining the bacterial cell wall.
The core structure of all beta-lactm antibiotics is the beta-lactam ring, a four-membered cyclic amide.
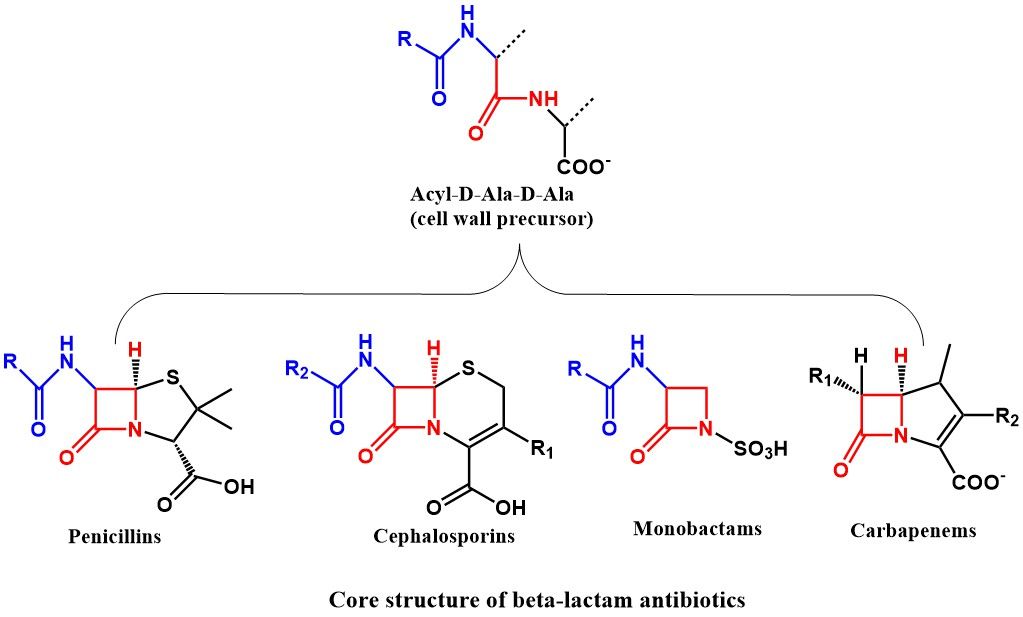
Mechanism of Inhibition:
- Binding to PBPs: Beta-lactams mimic the D-Ala-D-Ala terminal of peptidoglycan precursors, allowing them to bind to PBPs irreversibly.
- Inhibition of Transpeptidation: By binding to PBPs, beta-lactams prevent the cross-linking of peptidoglycan strands.
- Weakened Cell Wall: The inhibition of transpeptidation results in a structurally compromised cell wall that cannot withstand osmotic pressure.
- Cell Lysis: Due to the weakened wall, the bacterium swells and bursts as water enters via osmosis, leading to bacterial death.
Apart from inhibiting PBPs, beta-lactams also trigger autolysins, bacterial enzymes that degrade peptidoglycan. Normally, autolysins function in cell wall turnover, but when beta-lactams disrupt new cell wall formation, these enzymes become unregulated and contribute to cell wall destruction, further accelerating bacterial death.
Beta-lactam antibiotics are highly selective for bacteria because human cells lack peptidoglycan. Since our cells do not possess PBPs or a bacterial cell wall, beta-lactams do not harm human cells.
Bacteria can develop resistance through several mechanisms:
- Beta-lactamase enzymes: These enzymes hydrolyze the beta-lactam ring, rendering the antibiotic inactive (e.g., penicillinase, cephalosporinase).
- Altered PBPs: Some bacteria (e.g., MRSA) produce PBPs with low affinity for beta-lactams.
- Efflux Pumps: Actively pump out the antibiotic before it reaches PBPs.
- Porin Mutations: Gram-negative bacteria can modify their outer membrane porins, preventing antibiotic entry.
Solution to Resistance:
- Beta-lactamase inhibitors (e.g., clavulanic acid, tazobactam) are combined with beta-lactams to inhibit bacterial enzymes.
- Newer beta-lactams (e.g., carbapenems) have enhanced stability against beta-lactamases.
Beta-lactam antibiotics work by inhibiting bacterial cell wall synthesis via blocking PBPs, leading to bacterial lysis. Their effectiveness is limited by bacterial resistance mechanisms, which are countered using beta-lactamase inhibitors and novel antibiotic designs.
